Division of Hepato/Billiary/ Pancreatic
Many Hepatitis B carriers receive prophylactic antiviral drugs (nucleos(t)ide analogues, NUCs) while undergoing cancer chemotherapy. The greatest fear for these patients is that once the prophylactic medication is stopped, their immunity may decline, causing the dormant Hepatitis B virus (HBV) to "wake up" (reactivate). This can lead to severe complications, including fulminant hepatitis. It is a tragedy when a patient successfully stabilizes their cancer through grueling chemotherapy, only to face an increased risk of death due to a preventable Hepatitis B relapse.
This prospective study, spanning ten years, tracked chemotherapy patients receiving prophylactic NUCs and revealed several life-saving findings regarding viral reactivation:
1. Identifying the Risk Factors Our research discovered that patients with the following characteristics have a significantly higher risk of "virological relapse" (viral load spike) or "clinical hepatitis relapse" (abnormal liver function index):
l Medication Choice: Patients who used specific potent antiviral drugs (such as TDF or TAF) for prophylaxis, or those who received anthracycline-containing chemotherapy (often referred to as "Red Devil" chemotherapy).
l Initial Viral Load: Patients who already had a higher Hepatitis B viral load (HBV DNA) before starting treatment.
l End-of-Treatment Index: Patients whose Hepatitis B surface antigen (HBsAg) levels remained greater than 100 IU/mL at the end of prophylactic treatment saw their relapse risk skyrocket by nearly 6 to 8 times.
2. The Fatal Link Between Relapse and Mortality This is the study's most critical warning: once a patient stops prophylactic antivirals after chemotherapy, if a Hepatitis B relapse or hepatitis flare-up occurs, their overall risk of death increases significantly by approximately 3.3 to 3.6 times. This means that controlling the Hepatitis B virus is not just about protecting the liver; it is a key factor in ensuring the successful completion of cancer treatment and improving overall survival rates.
3. Implications for Patients and Physicians For high-risk groups (such as those with high viral loads or specific medication histories), standard preventive measures may not be enough. Medical teams need to monitor viral loads and antigen indices more frequently and consider more aggressive preventive strategies. Preventing Hepatitis B relapse is essentially protecting the patient's lifeline during their battle against cancer.
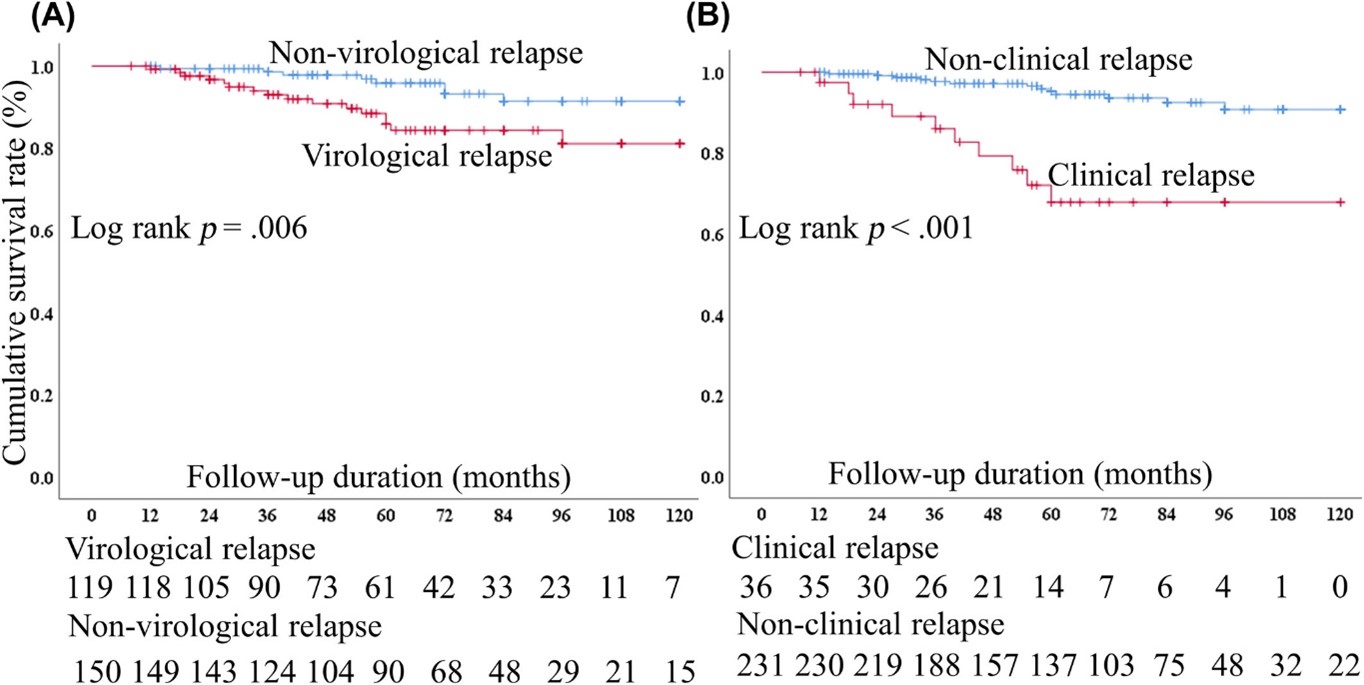
Figure 1. Compared cumulative survival rates by (A) virological and (B) clinical relapse in HBV patients receiving cytotoxic chemotherapy with prophylactic nucleos(t)ide analogues therapy.
Key Statistical Evidence
The overall 5-year cumulative survival rate for patients in this study was 91.0%, with an average survival time of 111.2 ± 1.7 months. Notably, patients who experienced "virological relapse" or "clinical relapse" had significantly lower cumulative survival rates compared to non-relapsers (Log-rank p=0.006 and p<0.001, respectively).
Multivariate analysis confirmed that after adjusting for age, gender, cancer stage, serum creatinine, and albumin, both virological relapse (Hazard Ratio: 3.32) and clinical relapse (Hazard Ratio: 3.59) are significant risk factors for all-cause mortality.
Wang C-W, Huang C-F, Yeh M-L, et al. Assessment of hepatitis B virus relapse in cancer patients receiving chemotherapy with prophylactic nucleos(t)ide analogues: Implications for overall mortality. Liver Int. 2024;44:2592-2604. doi:10.1111/liv.16030
